Based on easy-to-use and validated protocols, the SkinEthic tissue models allow industrial corporations to screen and measure with high precision, the irritation, penetration, metabolism, or efficacy of large number of formulations or actives, to eliminate those that are unsuitable, so that the final (in vivo) tests can be carried out on a selected few.
MULTIPLE END-POINT TESTING APPROACH
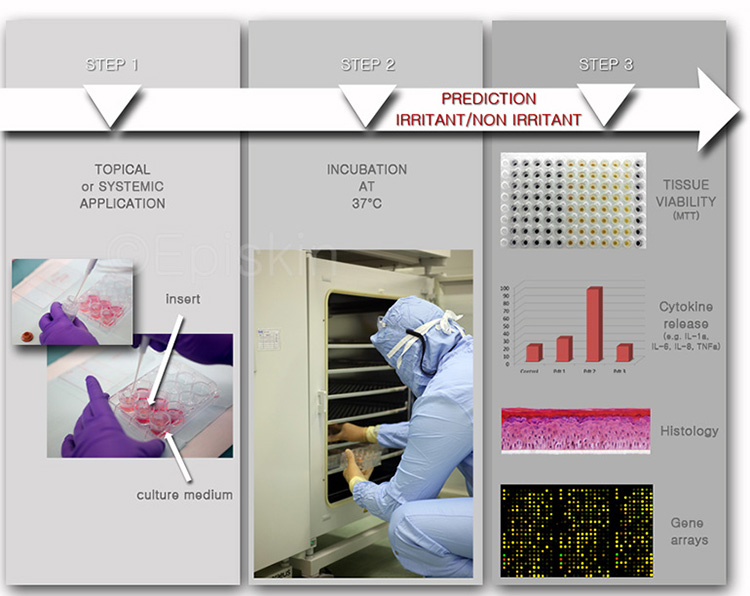
A small amount (2-10 mg/cm²) of test product (and controls) is deposited onto the surface of the tissues and spread with a small paintbrush. The cultures are incubated at 37°C for a specific time (from 10 minutes up to 6 days), after which they are analyzed for tissue histology, viability and the release of inflammatory mediators or cytokines (multiple end-point analysis).
MULTIPLE SAFETY SCREENING
